Blog
July 11, 2025
How to Write a Software Requirements Specification (SRS) Document
Application Lifecycle Management,
Software Quality
A detailed Software Requirements Specification (SRS) document is key to building software that meets both business needs and user expectations. Clear, concise, and executable requirements align project teams, offer clarity on functionality, and act as a single source of truth throughout development.
Whether you're using agile, waterfall, or a hybrid approach, this guide will help you craft clear, complete, and testable requirements. We'll discuss best practices and the benefits of using a dedicated requirements management tool to create your SRS vs. using Microsoft Word. We also include an example of an SRS document created in Perforce ALM.
▶️ See an SRS Document in Action
Back to top
What Is an SRS Document?
An SRS document acts as a guide for the development team, detailing how the software should perform and interact. It outlines the purpose, behavior, and functionality of a software application.
Think of an SRS as a blueprint or roadmap for the software you're going to build. The elements that comprise it should include:
- Purpose: Why the software is being built.
- Description: What the software will do.
- Requirements: Specific functionalities and constraints.
- Links: Connecting user stories to specific requirements.
- Approval Process: Ensuring all stakeholders are aligned.
Below is an example of a simple SRS document created using Perforce ALM:
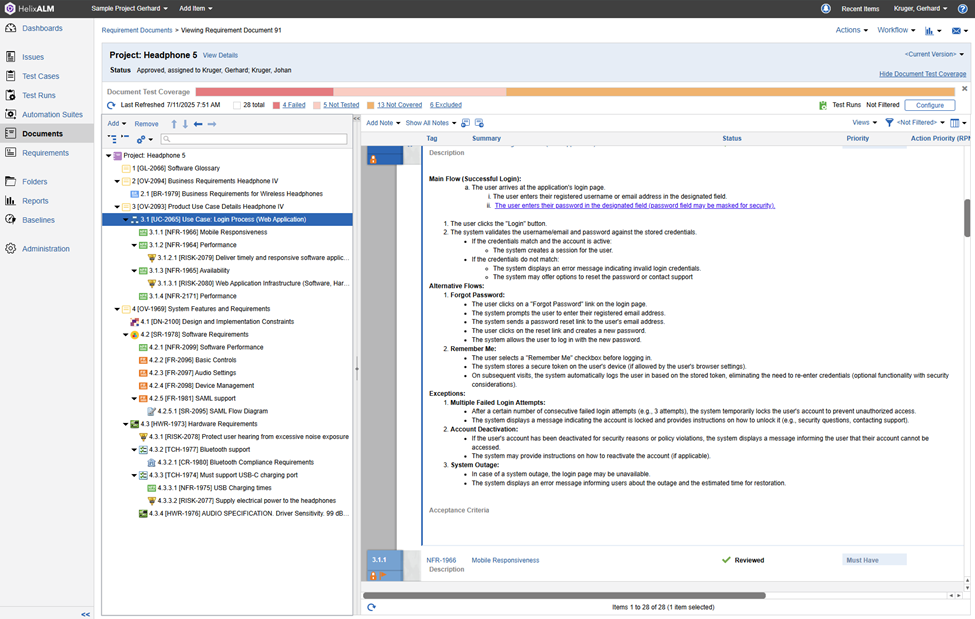
Who Uses an SRS?
Here is a breakdown of team member roles in producing and using an SRS:
- Product Owners and Business Analysts: Define requirements.
- Developers: Understand what to build.
- Testers: Create test cases and ensure validation.
- Project Managers: Track scope and progress.
- Clients and Stakeholders: Approve features and expectations.
Why Use an SRS Document?
A well-constructed SRS document provides everyone involved in your software development process with clear direction. It informs vital decisions along your product’s life cycle and ensures that each requirement is met.
It takes time and careful consideration to create a proper SRS, but the effort pays back in the development phase. The better your team understands your product and its users’ needs, the less time it will take to complete.
The Benefits of Using an SRS Document
Taking the time to properly draft a software requirements specification document:
- Improves Clarity: Removes ambiguity using controlled language.
- Sets Clear Expectations: Defines what success looks like.
- Boosts Traceability: Links requirements to design, code, and tests.
- Facilitates Testing: Ensures all features can be validated.
- Reduces Rework: Prevents scope creep by addressing potential issues upfront.
- Enables Compliance: Traceable, version-controlled requirements help meet regulatory standards.
How to Write an SRS Document: 7 Steps
1. Create an Outline or Use an SRS Template
Your first step is to create an outline for your software requirements specification. This may be something you create yourself, or you can use an existing SRS template.
If you’re creating the outline yourself, it may look something like this:
1. Introduction
1.1 Purpose
1.2 Intended Audience
1.3 Intended Use
1.4 Product Scope
1.5 Definitions and Acronyms
2. Overall Description
2.1 User Needs
2.2 Assumptions and Dependencies
3. System Features and Requirements
3.1 Functional Requirements
3.1.1 Numbered and Described
3.1.2 EARS format: "When [event], the system shall [response]")
3.1.3 Include Specification by Example or BDD format (e.g., Gherkin)
3.2 Non-Functional Requirements
3.2.1 Performance (e.g., "95% of requests shall return in under 2 seconds")
3.2.2 Security (e.g., "Only authenticated users can access admin API")
3.2.3 Usability, Reliability, Compliance
3.3 External Interface Requirements
3.3.1 Performance Requirements
3.3.2 Safety Requirements
3.3.3 Security Requirements
3.3.4 Software Quality Attributes
3.3.5 Business Rules
3.4 System Features
4. Other Requirements
4.1 Database Requirements
4.2 Legal and Regulatory Requirements
4.3 Internationalization and Localization
4.4 Risk Management (FMEA Matrix)
5. Appendices
5.1 Glossary
5.2 Use Cases and Diagrams
5.3 To Be Determined (TBD) List
This is a basic outline and yours may contain more (or fewer) items.
Download a white paper on best practices for writing requirements >>
2. Define your Product’s Purpose
Start by clearly defining why the software is being built and what it aims to achieve. This introduction sets expectations that you’ll revisit throughout the SRS.
Some items to keep in mind when defining this purpose include:
Intended Audience and Use
Define who in your organization will have access to the SRS and how they should use it. This may include developers, testers, and project managers. It could also include stakeholders in other departments, including leadership teams, sales, and marketing. Defining this now will lead to less work in the future.
Product Scope
Explain the broader objectives and benefits of the software. How does it align with your organization’s goals?
Definitions and Acronyms
Clearly define all key terms, acronyms, and abbreviations used in the SRS. This will help eliminate any ambiguity and ensure that all parties easily understand the document.
If your project contains a large quantity of industry-specific or ambiguous terminology or acronyms, you may consider including a project glossary.
>> Need to create a PRD? Here's a how-to with examples >>
3. Describe What You're Building
Give a high-level perspective of the software: its users, interactions, functions, and assumptions about its environment. Questions to address include:
- Why is this product needed?
- Who is it for?
- Is it a new product?
- Is it an add-on to a product you’ve already created?
- Is this going to integrate with another product?
Answering these questions early on aligns teams and accelerates production later.
User Needs
Describe who will use the product and how. Are they primary or secondary users? What is their role within their organization? What problem does the product solve for them?
Do you need to know about the purchaser of the product as well as the end user? If you're developing medical devices and med device software, you may also need to know the needs of the patient.
Assumptions and Dependencies
What does the software rely on to function properly (e.g., existing technologies or third-party integrations)? Take stock of these technical assumptions to better understand where your product might falter or fail. Understating and laying out these assumptions ahead of time will prevent headaches later.
Finally, you should note if your project is dependent on any external factors. For example: If you are reusing software elements from a previous project, the new project would depend on those operating correctly.
4. Detail Your Specific Requirements
For your development team to meet the requirements properly, include as much detail as possible. Consider breaking down your requirements into categories:
Functional Requirements
Functional requirements outline the software’s specific functionality, such as user authentication or data processing. These requirements benefit greatly from structured phrasing (like EARS) or format-driven clarity (like Gherkin or BDD). For example: rather than simply stating, "Users can log in," a requirement might say: "Given a valid username and password, when a user attempts to log in, then they are directed to their dashboard."
Within medical devices especially, these functional requirements may have a subset of domain-specific requirements.
External Interface Requirements
External interface requirements are specific types of functional requirements that outline how your product will interface with other components. These are especially important when working with embedded systems.
There are several types of interfaces you may have requirements for, including:
- User
- Hardware
- Software
- Communications
System Features
System features, like performance metrics or error handling, are a type of functional requirements that highlight what a system needs to function.
Non-Functional Requirements
Nonfunctional requirements, which help ensure that a product will work the way users and other stakeholders expect it to, can be just as important as functional ones.
These may include:
- Performance requirements
- Safety requirements
- Security requirements
- Usability requirements
- Scalability requirements
The importance of non-functional requirements may vary depending on your industry. To prove regulatory compliance, industries such as medical technology, life sciences, and automotive, must carefully track and manage detailed non-functional requirements.
IEEE provides guidance for writing software requirements specifications, if you’re a member.
Additional Resource: Requirements Engineering: 9 Best Practices with Examples
5. Assess Risk Using FMEA
Incorporate a structured process to assess potential failure modes within the system. Failure Modes and Effects Analysis (FMEA) is a systematic methodology used to identify potential failure modes, evaluate their causes and effects, and prioritize them based on severity, occurrence, and detectability.
These factors are combined to calculate a Risk Priority Number (RPN), using the formula:
RPN = Severity × Occurrence × Detection
Higher RPN values indicate greater risk and should be addressed with mitigation actions as early as possible.
Including an FMEA table or matrix in your SRS ensures that you address critical failure points early in the design process, minimizing the likelihood of system malfunctions. Here’s an example FMEA table to help you get started: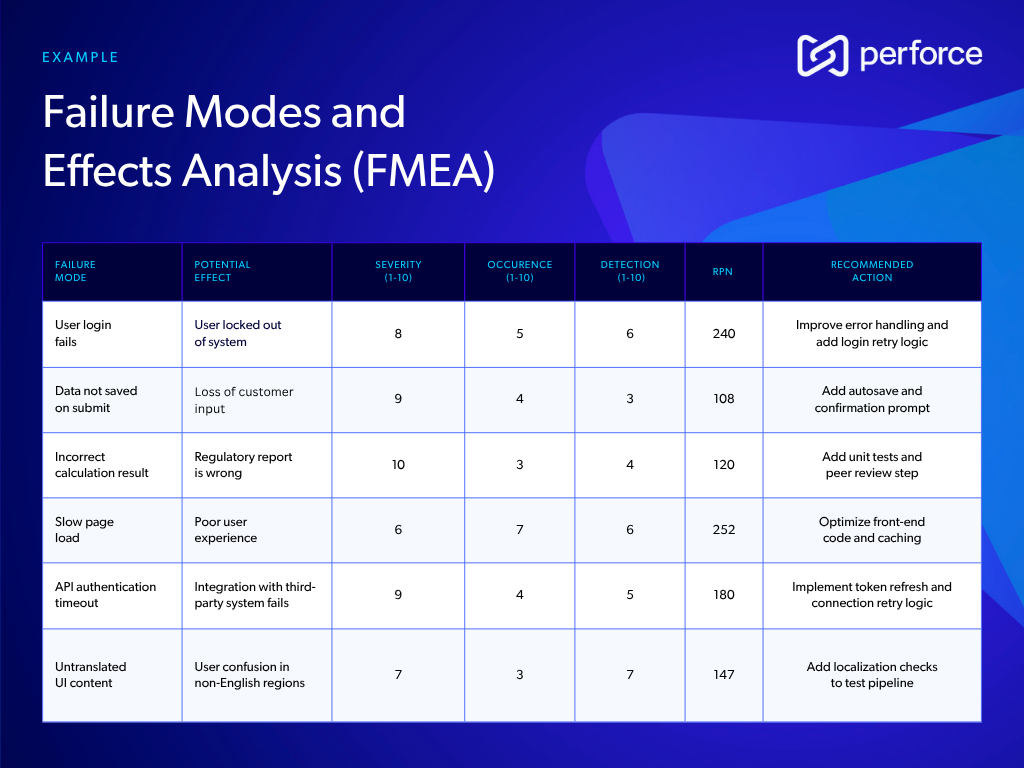
6. Link User Stories and Tasks to High-Level Requirements
Connecting user stories and tasks to high-level requirements ensures traceability throughout your development lifecycle. Map each user story to a specific requirement within the SRS to clarify its purpose and align it with project goals. To ensure consistency and avoid miscommunication, use clear identifiers in your links.
7. Deliver and Refine
Once your SRS document is drafted, circulate it for feedback and approval among stakeholders. An SRS is a living document; updates are often necessary as requirements evolve during development.
By adopting this approach, teams can more effectively track progress, prioritize work, and quickly identify gaps or misalignments. Tools such as requirement traceability matrices or software development management platforms can be used to facilitate this process, making it easy to link, monitor, and adjust user stories and tasks throughout the project lifecycle.
Back to topEvaluating Requirements Management Solutions?
Get our Requirements Management Software Buyer's Guide to learn 5 questions to ask when comparing traceability and compliance solutions.
10 Best Practices for Defining Software Requirements
To ensure your SRS is not only readable but actionable, follow these 10 dos and don'ts:
- Be Clear and Specific: Use concise and unambiguous language to ensure all stakeholders easily understand your requirements.
- Incorporate Specification by Example (SBE): Including realistic scenarios helps stakeholders understand exactly what the system should do.
- Don't Overload with Irrelevant Details: Focus on essential requirements and leave out extraneous information that may distract or confuse readers.
- Ensure Traceability: Map each requirement to design elements, test cases, and implementation components for easier debugging downstream.
- Include Acceptance Criteria: Define measurable criteria for each requirement to confirm success upon completion.
- Prioritize Requirements: Organize your requirements by importance to help your team focus on delivering high-value features first.
- Use Visual Aids: Diagrams, flowcharts, and tables illustrate complex ideas and enhance understanding across teams.
- Maintain Consistency: Use consistent structure, terminology, and formatting throughout your document to improve readability and prevent confusion.
- Review, Update, and Validate: Regularly review the document with all stakeholders to ensure accuracy, completeness, and alignment with project goals.
- Leverage Version Control and Collaboration Tools: Using ALM platforms ensures that changes are tracked and that collaboration doesn’t introduce inconsistencies.
Writing an SRS in Microsoft Word vs. Requirements Management Software
While tools like Microsoft Word or Google Docs make it simple to create a reuseable SRS document template, managing complex projects with these can lead to version control issues and inefficiencies. As requirements evolve, your SRS can quickly fall out-of-date, leading to frustrated teams and unnecessary rework.
For scalable projects, dedicated requirements management software like Perforce ALM, Jama and others provide significant advantages:
- Real-time updates prevent versioning conflicts.
- Traceability tools link requirements to testing and development phases.
- Faster reviews and approvals streamline timelines.
Opting for advanced tools to manage your SRS creates fewer delays and a higher-quality final product.
Back to topThe Value of a Dedicated SRS Tool
A well‑written Software Requirements Specification gives teams a shared understanding of what needs to be built, why it matters, and how success will be measured. When done right, it reduces ambiguity, prevents scope creep, and improves collaboration across product, development, and testing teams.
But writing the SRS is only the beginning.
As projects evolve, requirements change. Stakeholders request updates. Dependencies emerge. Teams need to review, approve, trace, and adapt requirements without losing alignment—or spending hours reworking static documents. This is where many teams struggle, especially when SRS documents live in spreadsheets or word processors that weren’t designed for ongoing collaboration.
A requirements management tool like Perforce ALM is a unified requirement and test management platform that streamlines and simplifies SRS workflows. Watch our full demo to see how you can link your requirements to test cases and trace test results back to requirements, for end-to-end traceability across the development process. For organizations in heavily regulated industries, this traceability helps you prove compliance and makes it easier to pass audits.
▶️ See an SRS Document in Action

